🩺 The Pulse: AI Workforce Tools and the Future of Cancer Screening
Plus: Finding trusted answers to clinical questions with Heidi Evidence
1. Triage – Your Fortnightly Rundown
Hi Pulse Readers - this week, we’re diving into:
how Patchwork Health’s AI rostering tool helps NHS hospitals reduce shift gaps and agency spending,
AI-prediction of breast cancer risk after an all-clear mammogram,
and how you can use Heidi Evidence to quickly review research and guidelines.
2. Case Study – Your Fortnightly Practical
Video Source: Hendrix Health
Using Heidi to Find Evidence
Case Presentation: Dr Harry have been using Ask Heidi to turn his completed ADHD assessment notes into ad-hoc documents when he doesn’t have a template, and it’s been working well.
Today, he is about to see a new patient who describes a long history of inattention, disorganisation, and poor follow through dating back to school, but the presentation is not straightfoward. They are now struggling at work, sleeping poorly, feeling anxious, and drinking alcohol regularly.
Dr Harry is not sure whether this is uncomplicated adult ADHD or another condition presenting in a similar way. He does not want to rely on memory alone or waste time seraching across multiple sources while the patient is in the waiting room.
He wants a fast way to review quality evidence, sense-check the differential, and identify what else he should ask before making any decisions.
Approach: Use Heidi Evidence when the clinical picture is unclear and you want a faster, more efficient way to review trusted sources, including local guidelines and research studies.
1. Enter the clinical question in plain language
Start by typing the scenario into Heidi Evidence as you would naturally think about it in practice. For example:
“I’m about to see a 25M, long history of inattention and disorganisation now affecting work, some anxiety, poor sleep, regular EtOH use, ?ADHD ?something else, what should i think about patient is in waiting room”
You do not need to write a perfect academic search query. A clinical shorthand is usually enough for Heidi Evidence to return a useful starting point.
2. Review cite sources and key points from Heidi Evidence
Heidi Evidence can help explore alternative explanations and common overlaps, in this case anxiety disorder, depression, sleep disorders, substance use, autism spectrum, and medical contributors. This is especially useful in cases where the symptoms are non-specific and the differentials are not yet clear.
If you use Heidi Evidence to review evidence, for example by asking, “What is the latest research on psychedelics in mental health treatment?”, Heidi Evidence would often generate a summarised answer with key points at the top, followed by a more detailed response. Each point is linked to its source, so you can quickly see where the information is coming from and open the original article or guideline if you wish to.
3. Ask focused follow-up questions to guide the consult
Once the initial answer appears, you can narrow the query further by asking follow-up questions. You can also use it like Ask Heidi, or link the question to a consultation or patient.
You can also upload your own custom sources, so Heidi’s answers are tailored to your hospital or practice environment, rather than generic evidence and guidelines.
4. Use the evidence to support, not replace, clinical judgement
Heidi Evidence does not make diagnostic recommendations. Instead, it gives you a fast and efficient way to review evidence and guidelines related to the clinical context.
You still need to review the output critically and apply clinical judgement before making decisions on further management.
Outcomes: Using Heidi Evidence, Dr Harry can approach a more complex consultation in an evidence-informed way. He can quickly review research studies and local guidelines by asking questions. This helps him practise more safely, and make clinical decisions with greater confidence when the presentationis not straightforward.
Disclaimer: Hendrix Health is the official New Zealand partner for Heidi Health.
3. The Pulse - Your Fortnightly Update
AI Rostering Tool Rolls Out Across NHS Trusts to Cut Shift Gaps and Agency Spend
(a February update worth revisiting)
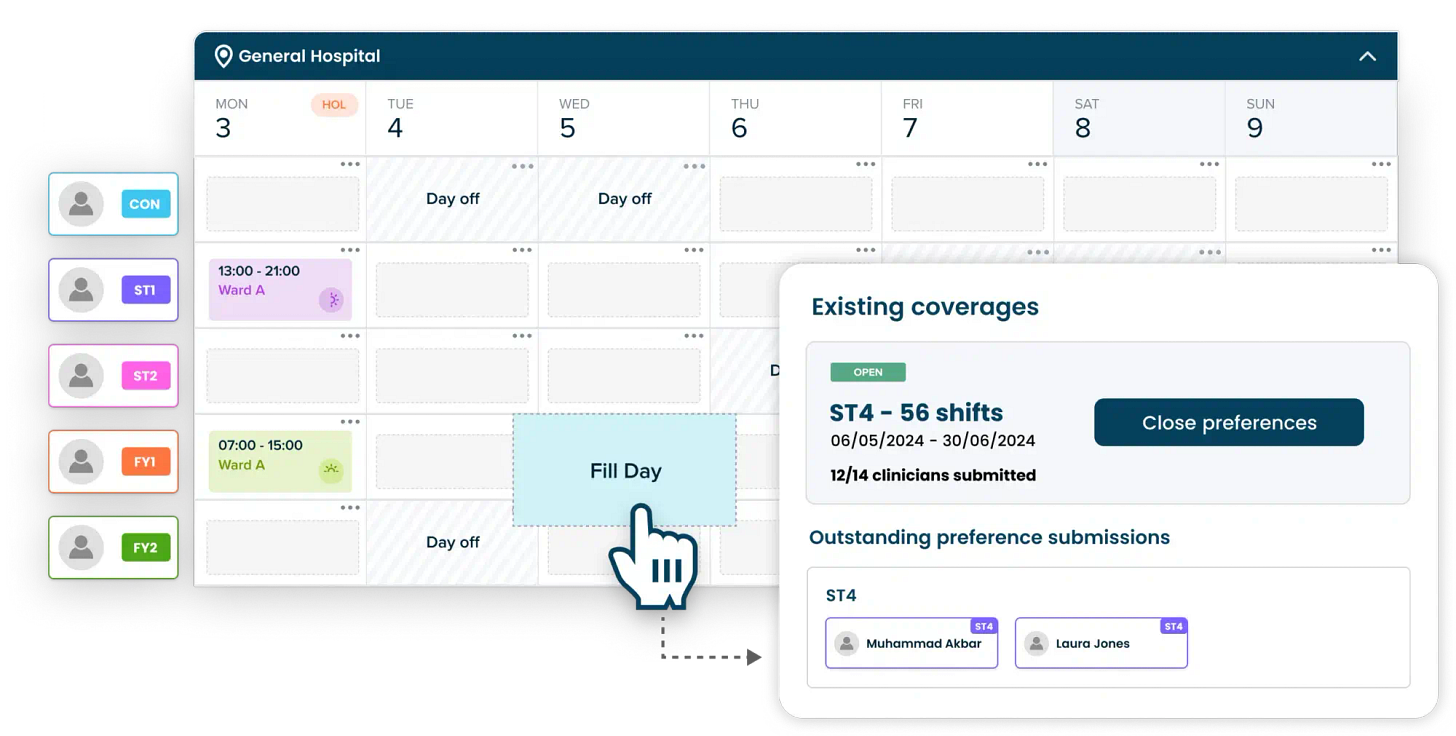
Patchwork Health, a doctor-led workforce management platform, has launched an AI-powered Preference-Based Rostering tool for NHS hospitals, designed to build clinician rotas that better match staff preferences while still meeting compliance and operational requirements. The tool is already being used at four NHS trusts across London, Yorkshire, and the South West of England.
Image Source: Patchwork Health
The system collects clinician shift preferences, alongside workforce rules, minimum staffing requirements, and service demand. Its AI algorithm then processes thousands of possible roster combinations to generate a flexible rota in minutes, distributing night shifts, weekend work, and longer shifts more fairly than traditional manual scheduling.
Once the rota is produced, managers spend less time making repeated manual changes after publication, while clinicians gain more control over their working patterns. This is intended to reduce rota-related frustration, cut shift gaps, and lower reliance on temporary agency staff, while keeping service delivery stable.
Key Features:
Operational impact: In an early 10-week ward trial comparing the tool with a traditional roster-building process, Patchwork reported a 97% reduction in unfilled shifts.
Cost reduction: Temporary staffing costs fell by 98%, from £18,000 to £400 over the same 10-week period.
Preference-based design: The system is built to account for both positive and negative clinician shift preferences, with Patchwork’s modelling suggesting rosters can meet 98% of negative preferences at the point of creation.
Established NHS footprint: Patchwork says it already works with over 200 healthcare organisations, is partnered with over half of NHS trusts and health boards across the UK, and has supported more than 120,000 healthcare professionals through its wider workforce platform.
Implications for the Health System and Clinicians:
This is one of the more practical healthcare AI rollout stories from recent weeks because it targets a real operational bottleneck. Rostering is a high-friction problem that affects clinician retention, burnout, staffing gaps, and agency spend.
The broader lesson is that some of the most useful AI in healthcare may come from workforce tools that make services easier to operate, not just diagnostic and clinical decision support. The obvious caution is that the strongest result so far come from early company-reported trials, so the real test will be whether these gains hold across full NHS rollouts at scale.
AI in Breast Screening: What the BRAIx Risk Score Means for Personalised Screening
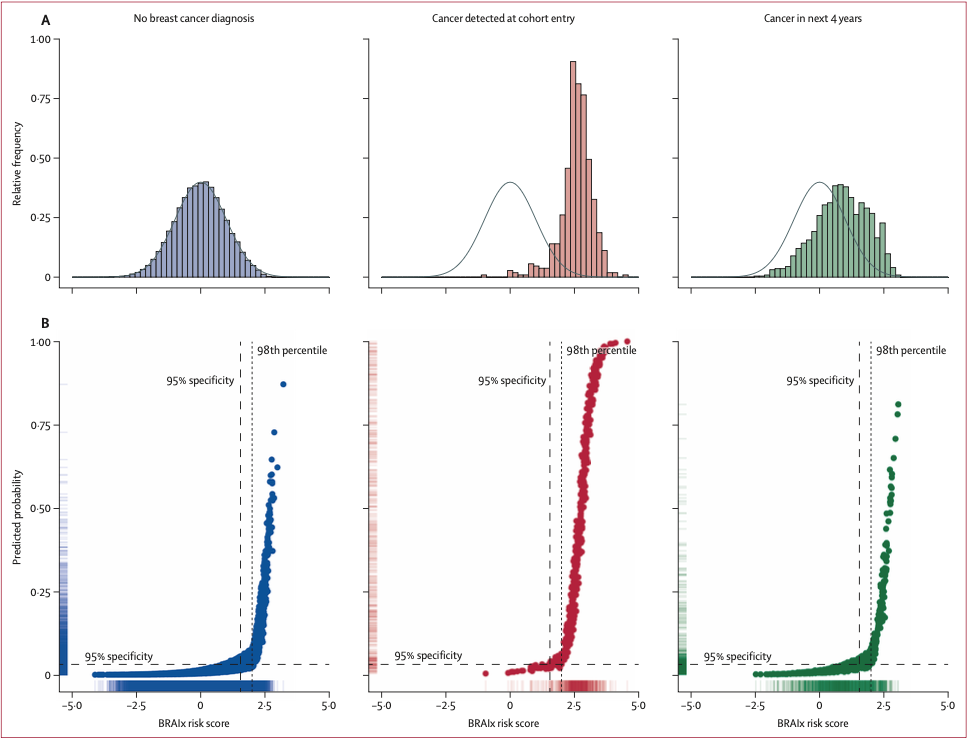
A population cohort study in The Lancet Digital Health evaluated the BRAIx risk score, an AI-derived mammography-based tool that was repurposed from detection, to the prediction of both current and 4-year breast cancer risk.
The study used a training dataset of nearly 400,000 women, then tested the model in an independent cohort of over 96,000 women from BreastScreen Victoria, Australia with external validation in a Swedish screening population.
Image Source: The Lancet Digital Health
Key Findings:
Strong intermediate-term risk prediction: The BRAIx risk score was a strong predictor of breast cancer diagnosed within 4 years after an all-clear screen, with consistent performance across both Australian and Swedish screening datasets.
High-risk subgroup identified despite an initial all-clear: Among the top 2% of women with the highest BRAIx risk scores who were initially cleared at screening, 9.7% were diagnosed with breast cancer within 4 years, a risk level higher than that reported for BRCA1 and BRCA2 mutation carriers over the same timeframe.
Outperformed traditional risk factors: The BRAIx risk score was a stronger predictor of 4-year breast cancer risk than age, family history, mammographic density, recall status, screening round, or country of birth. After accounting for BRAIx, mammographic density was no longer a significant predictor in the Australian test data.
The score alone was as good as full risk models: Full multivariable models performed only marginally better than the BRAIx risk score alone, suggesting that traditional risk factors contribute little additional predictive value once the AI score is already accounted for.
Implications for Healthcare Systems:
For screening programmes, the practical implication is a shift toward more personalised, risk-based pathways, such as closer follow-up or supplemental imaging for women at highest intermediate-term risk, rather than a one-size-fits-all screening model. The caution, however, is important: this was still a risk-prediction study, not a prospective implementation trial, so any policy change will still need real-world validation, cost-effectiveness modelling, and careful attention to equity.
Read the full study here.
4. Vitals – Quick Bytes
New Zealand explores AI Integration for Breast Screening
Health New Zealand is exploring how AI can safely support BreastScreen Aotearoa, with current scoping extending beyond mammogram reading to breast density reporting, image quality support at acquisition, and breast cancer risk stratification. The move comes as screening demand rises, radiologist workforce shortages persist, the screening age extends to 74, and transition from largely paper-based, opt-in process to a digital-first, opt-out register through Te Puna. International evidence, including Sweden’s MASAI trial, suggests AI-supported screening may improve early cancer detection and reduce interval cancers. There is a clear opportunity for New Zealand, but the real challenge is implementation, ensuring any adoption is safe, equitable, and workable within the existing infrastructure. (healthcareitnews.com)
UK Pilots AI and Robotic Bronchoscopy to Speed Lung Cancer Diagnosis
The NHS has launched a pilot at Guy’s and St Thomas’ that combines Optellum’s AI risk stratification for low-dose CT lung scans with Intuitive’s Ion robotic bronchoscopy to identify and biopsy small, high-risk lung nodules, including lesions as small as 6 mm. The aim is to shorten diagnosis by replacing weeks of repeat scans and invasive procedures with a single 30-minute biopsy appointment as targeted lung screening expands across England. More than 300 robotic biopsy procedures have already been carried out, with 215 patients going on to receive cancer treatment. The main caution is that this remains a pilot, so impacts on waiting times, outcomes, cost-effectiveness, bias, and equity still need full evaluation before wider rollout.
We’d love to hear your thoughts, so join the conversation by leaving a comment below:
Stay tuned for more insights in the next edition of The Pulse.
Have a great day & see you in two weeks!
Your Hendrix Health Team